Event Details
*** The 11th Annual Zane’s Run will be held on Sunday, September 29, 2019 at:
Great Valley Middle School
255 North Phoenixville Pike, Malvern, Pa 19355***Day of Registration opens: 7:30 a.m.
Fun Run: 9:00 a.m.
5k Run: 9:15 a.m.
Race Winners Announced: 10:15 a.m.*** Walk up registrations welcome the day of the race beginning at 7:30 a.m. Cash, check, & credit cards will be accepted for registrations. Please Note- you are not guaranteed a t- shirt/ medal for any registrations AFTER August 30th!
***Early bird registration gets you $5 off per registration and an event t-shirt.
*** Chip Timing!
*** Zane’s Run will be held Rain or Shine!!!
Proceeds go to Cure SMA
Our goal is to bring awareness about Spinal Muscular Atrophy (SMA) as well as to purchase the necessary medical adaptive equipment for children with Type I and Type 2 SMA. Additionally, SBZ purchases & sends international SMA information CD’s for newly diagnosed families.
Find Us on Facebook
Find & join our Sweet Baby Zane group on facebook.
Additional Questions?
Please email zanesrun@gmail.com.
Treatments for SMA
Thanks to the dedication of our community and the ingenuity of our researchers, we now have multiple approved treatments that target the underlying genetics of SMA. But our work is not done. We know what we need to do to develop and deliver effective therapies. And we’re on the verge of further breakthroughs that will continue to change the course of SMA for everyone affected—from infants to adults—and eventually lead to a cure.
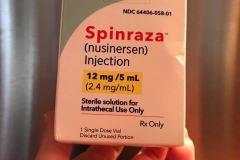
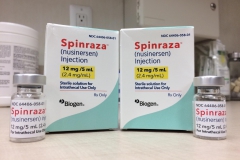
On December 23, 2016, the FDA announced that it had approved Spinraza, a treatment developed by Biogen and Ionis, making it the first-ever approved therapy for SMA. Cure SMA provided the very first research funding for this program beginning in 2003.
On May 24, 2019, the FDA announced that it had approved Zolgensma, a one-time intravenous (IV) gene therapy, to treat SMA. Zolgensma is approved for patients with all types and forms of SMA who are under two years of age at the time of dosing. Cure SMA supported this program with a series of grants to Nationwide Children’s Hospital, beginning in 2010.
Learn More:






 Find Us Now
Find Us Now